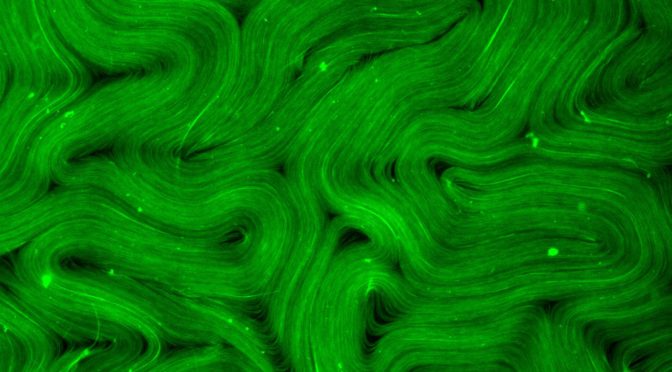
This work examines self-mixing in active nematics, a class of fluids in which mobile topological defects drive chaotic flows in a system comprised of biological filaments and molecular motors. We present experiments that demonstrate how geometrical confinement can influence the braiding dynamics of the defects. Notably, we show that confinement in cardioid-shaped wells leads to realization of the golden braid, a maximally efficient mixing state of exactly three defects with no defect creation or annihilation. We characterize the golden braid state using different measures of topological entropy and the Lyapunov exponent. In particular, topological entropy measured from the stretching rate of material lines agrees well with an analytical computation from braid theory. Increasing the size of the confining cardioid produces a transition from the golden braid, to the fully chaotic active turbulent state.
Tag Archives: featured
Colloidal aggregation in liquid crystal
Colloidal aggregation in anisotropic liquid crystal solvent
Devika G. Sudha, Jocelyn Ochoa and Linda S. Hirst, Soft Matter (2021) https://doi.org/10.1039/D1SM00542A
The mutual attraction between colloidal particles in an anisotropic fluid, such as the nematic liquid crystal phase, leads to the formation of hierarchical aggregate morphologies distinct from those that tend to form in isotropic fluids. Previously it was difficult to study this aggregation process for a large number of colloids due to the difficulty of achieving a well dispersed initial colloid distribution under good imaging conditions. In this paper, we report the use of a recently developed self-assembling colloidal system to investigate this process. Hollow, micron-scale colloids are formed in situ in the nematic phase and subsequently aggregate to produce fractal structures and colloidal gels, the structures of which are determined by colloid concentration and temperature quench depth through the isotropic to nematic phase transition point. This self-assembling colloidal system provides a unique method to study particle aggregation in liquid crystal over large length scales. We use fluorescence microscopy over a range of length scales to measure aggregate structure as a function of temperature quench depth, observe ageing mechanisms and explore the driving mechanisms in this unique system. Our analyses suggest that aggregate dynamics depend on a combination of Frank elasticity relaxation, spontaneous defect line annihilation and internal aggregate fracturing.
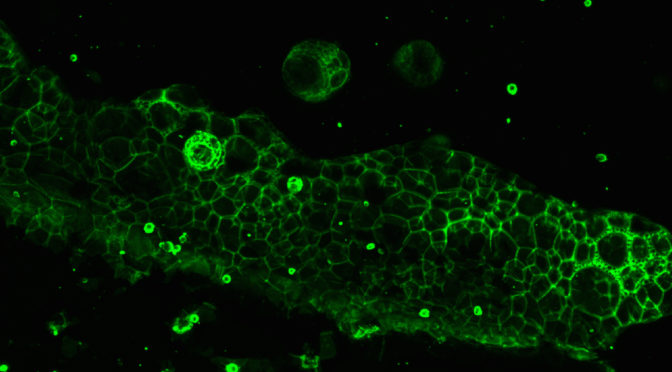
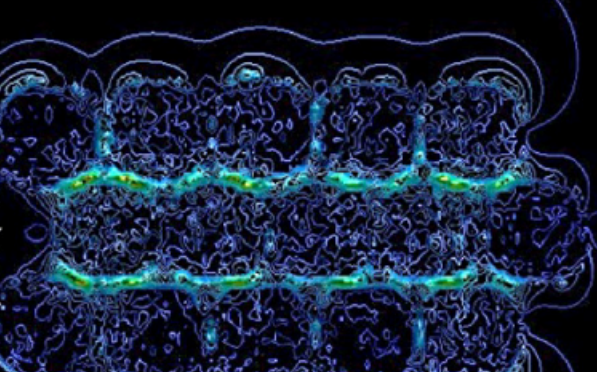
Submersed micropatterned structures control active nematic flow
Submersed micropatterned structures control active nematic flow, topology, and concentration
Kristian Thijssen*, Dimitrius A. Khaladj*, S. Ali Aghvami, Mohamed Amine Gharbi, Seth Fraden, Julia M. Yeomans, Linda S. Hirst, and Tyler N. Shendruk
PNAS September 21, 2021 118 (38) e2106038118; https://doi.org/10.1073/pnas.2106038118
Coupling between flows and material properties imbues rheological matter with its wide-ranging applicability, hence the excitement for harnessing the rheology of active fluids for which internal structure and continuous energy injection lead to spontaneous flows and complex, out-of-equilibrium dynamics. We propose and demonstrate a convenient, highly tunable method for controlling flow, topology, and composition within active films. Our approach establishes rheological coupling via the indirect presence of fully submersed micropatterned structures within a thin, underlying oil layer. Simulations reveal that micropatterned structures produce effective virtual boundaries within the superjacent active nematic film due to differences in viscous dissipation as a function of depth. This accessible method of applying position-dependent, effective dissipation to the active films presents a nonintrusive pathway for engineering active microfluidic systems.
Merced team studying shark electrosensing
Our lab recently published two new papers on the subject of electro-sensing in cartilaginous fishes, a group that includes sharks and rays. The sensing organ (AoL) in these fish is filled with a gel-like substance and we are interesting is figuring out how this gel works!
In the first paper our team, Lead by Molly Phillips, a graduate student in Chris Amemiya’s lab at Merced investigated the role of chitin in the electrosensory gel.
“Evidence of chitin in the ampullae of Lorenzini of chondrichthyan fishes”Molly Phillips, W. Joyce Tang, Matthew Robinson, Daniel Ocampo Daza, Khan Hassan, Valerie Leppert, Linda S. Hirst, Chris T. Amemiya, , Current Biology, Volume 30, Issue 20, 2020, Pages R1254-R1255, https://doi.org/10.1016/j.cub.2020.08.014.
The second paper focuses on the structure of the gel.
Structural Characteristics and Proton Conductivity of the Gel Within the Electrosensory Organs of Cartilaginous Fishes Molly Phillips, Alauna Wheeler, Matthew J Robinson, Valerie Leppert, Manping Jia, Marco Rolandi, Linda S Hirst, Chris T Amemiya, ISCIENCE, in press (2021) August 03, (2021) https://doi.org/10.1016/j.isci.2021.102947
Co-authors include Physics graduate student Alauna Wheeler, who conducted the X-ray work with Phillips at the Advanced Light Source at Lawrence Berkeley National Laboratory; graduate student Matthew Robinson; and UC Santa Cruz electrical engineering Professor Marco Rolandi and his graduate student Manping Jia. The Santa Cruz team provided equipment and expertise for the conductance measurements.
Topological Chaos in active nematics
Topological chaos in active nematics, Amanda J. Tan, Eric Roberts, Spencer Smith, Ulyses Alvarado, Jorge Arteaga, Sam Fortini, Kevin Mitchell, and Linda S. Hirst, Aug 5th, NATURE PHYSICS (2019) Link
Abstract: Active nematics are out-of-equilibrium fluids composed of rod-like subunits, which can generate large-scale, self-driven flows. We examine a microtubule-kinesin-based active nematic confined to two dimensions, exhibiting chaotic flows with moving topological defects. Applying tools from chaos theory, we investigate self-driven advection and mixing on different length scales. Local fluid stretching is quantified by the Lyapunov exponent. Global mixing is quantified by the topological entropy, calculated from both defect braiding and curve extension rates. We find excellent agreement between these independent mea-sures of chaos, demonstrating that the extensile stretching between microtubules directly translates into macroscopic braiding of positive defects. Remarkably, increasing extensile activity (through ATP concentration) does not increase the dimensionless topological entropy. This study represents an application of chaotic advection to the emerging field of active nematics and quantification of the collective motion of an ensemble of defects (through topological entropy) in a liquid crystal.
The full text of the paper can be found here https://rdcu.be/bM3Gi

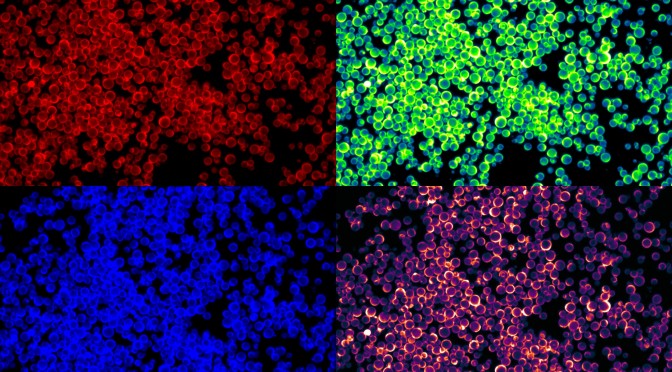
Nanofoams – Nature communications
Nanoparticle-based hollow microstructures formed by two-stage nematic nucleation and phase separation
Sheida T. Riahinasab, Amir Keshavarz, Charles N. Melton, Ahmed Elbaradei, Gabrielle I. Warren, Robin L. B. Selinger, Benjamin J. Stokes & Linda S. Hirst
Nature Communications volume 10, Article number: 894 (2019)
Rapid bulk assembly of nanoparticles into microstructures is challenging, but highly desirable for applications in controlled release, catalysis, and sensing. We report a method to form hollow microstructures via a two-stage nematic nucleation process, generating size-tunable closed-cell foams, spherical shells, and tubular networks composed of closely packed nanoparticles. Mesogen-modified nanoparticles are dispersed in liquid crystal above the nematic-isotropic transition temperature (TNI). On cooling through TNI, nanoparticles first segregate into shrinking isotropic domains where they locally depress the transition temperature. On further cooling, nematic domains nucleate inside the nanoparticle-rich isotropic domains, driving formation of hollow nanoparticle assemblies. Structural differentiation is controlled by nanoparticle density and cooling rate. Cahn-Hilliard simulations of phase separation in liquid crystal demonstrate qualitatively that partitioning of nanoparticles into isolated domains is strongly affected by cooling rate, supporting experimental observations that cooling rate controls aggregate size. Microscopy suggests the number and size of internal voids is controlled by second-stage nucleation.
A plasmon induced liquid crystal device
“All optical switching of nematic liquid crystal films driven by localized surface plasmons” M.T. Quint, S. Delgado, Z.S. Nuno, L.S. Hirst and S. Ghosh, OPTICS EXPRESS, 23,5, 6888 (2015) Link
We have demonstrated an all-optical technique for reversible in-plane and out-of-plane switching of nematic liquid crystal molecules in few micron thick films. Our method leverages the highly localized electric fields (“hot spots”) and plasmonic heating that are generated in the near-field region of densely packed gold nanoparticle layers optically excited on-resonance with the localized surface plasmon absorption. Using polarized microscopy and transmission measurements, we observe this switching from homeotropic to planar over a temperature range starting at room temperature to just below the isotropic transition, and at on-resonance excitation intensity less than 0.03 W/cm2. In addition, we controllably vary the in-plane directionality of the liquid crystal molecules in the planar state by altering the linear polarization of the incident excitation. Using discrete dipole simulations and control measurements, we establish spectral selectivity in this new and interesting perspective for photonic application using low light power.
Quantum dot micro-shells
“Self-assembled nanoparticle micro-shells templated by liquid crystal sorting”A. R. Rodarte, B.H. Cao, H. Panesar, R.J. Pandolfi, M. Quint, L. Edwards, S. Ghosh, J.E. Hein and L.S. Hirst, Soft Matter, 10.1039/C4SM02326A (2015) Link
A current goal in nanotechnology focuses on the assembly of different nanoparticle types into 3D organized structures. In this paper we report the use of a liquid crystal host phase in a new process for the generation of micron-scale vesicle-like nanoparticle shells stabilized by ligand–ligand interactions. The constructs formed consist of a robust, thin spherical layer, composed of closely packed quantum dots (QDs) and stabilized by local crystallization of the mesogenic ligands. Ligand structure can be tuned to vary QD packing within the shell and made UV cross-linkable to allow for intact shell extraction into toluene. The assembly method we describe could be extended to other nanoparticle types (metallic, magnetic etc.), where hollow shell formation is controlled by thermally sorting mesogen-functionalized nanoparticles in a liquid crystalline host material at the isotropic to nematic transition. This process represents a versatile method for making non-planar 3D nano-assemblies.
Congratulations Dr Pandolfi!
Congratulations go to Ron Pandolfi, the latest PhD graduate from the Hirst group. Ron’s PhD defense was on Monday Dec 8th, where he presented his thesis on “Self-assembly and Design of Tunable Soft Materials”
During his time in the lab Ron’s work has included molecular dynamics simulations of semi-flexible polymers and x-ray characterization of different soft systems.
Ron was recently hired at the Advanced Light Source, Lawrence Berkeley National Lab in Berkeley, CA where he’ll be working with soft matter x-ray team.
Magnetic field induced quantum dot brightening
The design and development of multifunctional composite materials from artificial nano-constituents is one of the most compelling current research areas. This drive to improve over nature and produce ‘meta-materials’ has met with some success, but results have proven limited with regards to both the demonstration of synergistic functionalities and in the ability to manipulate the material properties post-fabrication and in situ. Here, magnetic nanoparticles (MNPs) and semiconducting quantum dots (QDs) are co-assembled in a nematic liquid crystalline (LC) matrix, forming composite structures in which the emission intensity of the quantum dots is systematically and reversibly controlled with a small applied magnetic field (<100 mT). This magnetic field-driven brightening, ranging between a two- to three-fold peak intensity increase, is a truly cooperative effect: the LC phase transition creates the co-assemblies, the clustering of the MNPs produces LC re-orientation at atypical low external field, and this re-arrangement produces compaction of the clusters, resulting in the detection of increased QD emission. These results demonstrate a synergistic, reversible, and an all-optical process to detect magnetic fields and additionally, as the clusters are self-assembled in a fluid medium, they offer the possibility for these sensors to be used in broad ranging fluid-based applications.